BOTOX IS POPULARLY USED IN TREATMENT:
- Correction of strabismus (strabismus) in patients 12 years of age and older.
- Treatment of spasmodic torticollis (cervical dystonia) in adults.
- Treatment of primary, severe, persistent axillary hyperhidrosis in adults that interfere with activities of daily living and are resistant to topical therapy.
- Relief of headache in adults with chronic migraine (headaches for at least 15 days/month of which at least 8 days are migraines).
- Temporarily improves the appearance of moderate to severe vertical lines between the eyebrows seen with frowning (known as interbrow lines), in adults under 65 years of age, when severe of these wrinkles have important psychological effects on the patient.

- Temporarily improves the appearance of moderate to severe nasolabial folds (crow’s feet wrinkles) in adults treated alone or concurrently with interbrow wrinkle.
- Temporarily improves the appearance of moderate to severe forehead wrinkles seen with maximal brow lift in adults
WHY NEED TO CHOOSE A PRESTIGE PLACE FOR BOTOX INJECTION TREATMENT?
BOTOX® is indicated:
- The recommended dose and frequency of BOTOX® administration should not be exceeded.
- The safe and effective use of BOTOX® depends on the proper storage of the drug, correct dose selection and correct mixing and administration techniques.
- Physicians should be knowledgeable of the anatomy involved and any anatomical changes resulting from previous surgical procedures prior to administration of BOTOX® and caution should be exercised when injecting into or near anatomic structures. surgery is vulnerable. Serious adverse events including fatal outcomes have been reported in patients who received BOTOX® injections directly into the salivary glands, oropharyngeal, esophagus, and stomach areas. Some patients have had pre-existing dysphagia or significant weakness. Injection procedure-related pneumothorax has been reported following injection of BOTOX® proximal to the chest. Care should be taken when injecting near the lungs, especially the apex.
- Care should be taken when using BOTOX® in the event of inflammation at the injection site or when there is excessive muscle weakness or atrophy in the intended muscles.
CONTRAINDICATIONS
BOTOX® is contraindicated for use:
a) in individuals with known hypersensitivity to any of the ingredients
of drugs;
b) in patients with severe myasthenia gravis or Eaton-Lambert syndrome.
BOTOX® is contraindicated when there is an infection at the injection site.
BOTOX® treats bladder dysfunction (overactivity of the bladder wall muscles
associated with neuropathy and overactive bladder) is also contraindicated for:
• The patient has an acute urinary tract infection.
• Patients with acute urinary retention who do not routinely perform self-catheterization
interval (CIC).
• The patient is unwilling or unable to have a post-treatment catheterization if required.
• The patient is having bladder stones.
PACKING

BOTOX® is supplied in clear glass vials containing sterile vacuum drying powder,
with a rubber stopper, tamper-proof aluminum sealing ring and transparent lid.
Box of 1 vial of 100 units.
STORAGE CONDITIONS
- Unopened vials should be stored in the refrigerator (2°-8°C).
- After reconstitution, BOTOX® can be stored in the refrigerator (2o-8oC) for 24 hours before use.
All vials, including expired vials, or used instruments that come into direct contact with the drug should be disposed of as medical waste. In the event that a toxin inactivation is desired (e.g. spillage), it is recommended to use a dilute solution of hypochlorite (0.5% or 1%) for 5 minutes prior to disposal as medical waste.
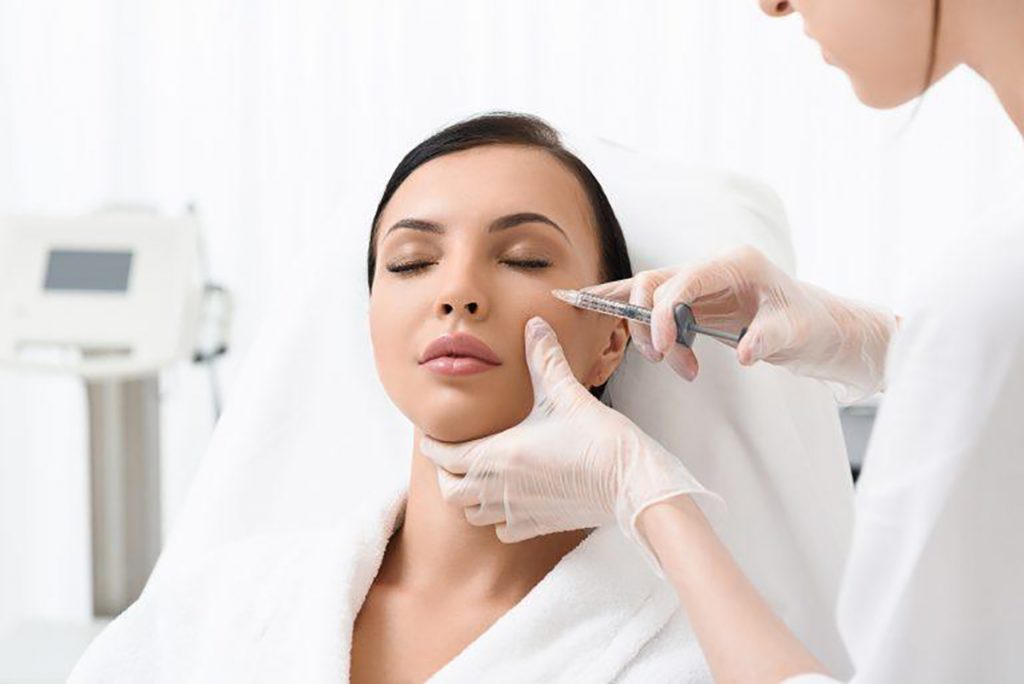
At Stamford Skin Center, we use BOTOX products with clear origin, customers will be checked by a doctor before performing treatment. Highly skilled doctors, ensuring the aesthetics of the face when treating with BOTOX injection method.